An original TRACK-TBI investigation shows that being discharged from a hospital or trauma center after receiving treatment for a TBI does not always mean that a patient is fully recovered. A nationwide study lead by clinicians and scientists at UCSF and University of Southern California, show that more than half of TBI patients do not receive the follow up care they need. <<Read More>>
News
March 27, 2018
Read more about the TED Initiative and research supporting a lifetime of brain injury on the U.S. Army website as part of Brain Injury Awareness month.
March 07, 2018
FOR IMMEDIATE RELEASE
FROM MARY ANN LIEBERT, INC., PUBLISHERS
December 08, 2017
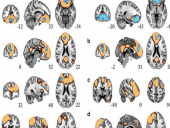
Dr. Pratik Mukjerjee's paper, “Resting-State Functional Connectivity Alterations Associated with Six-Month Outcomes in Mild Traumatic Brain Injury” was the 2nd most-downloaded paper in the Journal of Neurotrauma in the last 12 months. Congratulations to Dr. Mukherjee and his team of co-authors. To download the paper, click here.