Medical Device Development Tools (MDDT)
The FDA's Medical Device Development Tools (MDDT) program is a way for the FDA to qualify tools that medical device sponsors can use in the development and evaluation of medical devices. Qualification means that the FDA has evaluated the tool and concurs with available supporting evidence that the tool produces scientifically-plausible measurements and works as intended within the specified context of use. The context of use depends on the product area, the stage of medical device development, and the role of the tool in device evaluation. The program promotes innovation in medical device development and regulatory science to help bridge the gap between research of medical devices and the delivery of devices to patients.
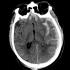
TED Initiative Receives FDA Qualification of Medical Device Development Tool
Dr. Esther Yuh and her TED Seed Project received FDA qualification of their Medical Device Development Tool submitted on behalf of the TED Initiative.
The OsiriX CDE Software Module, is only the third qualification of a medical device development tool (MDDT) by the FDA, the first of a biomarker test tool of any type, and the first such tool for TBI. A biomarker test is a lab test or instrument used to detect or measure an indicator of biologic processes or pharmacologic responses to a treatment (biomarker). This qualification provides a tool for more efficient development of devices in a critical area of medicine – traumatic brain injury (TBI) treatment. <<ReadMore>>